Abiotic and biotic stresses are one of the major threats to food security worldwide and their severity is expected to increase under the projected global warming. As plants cannot escape from unfavourablegrowth conditions, they must continuously cope with these stress factors in the most effective and energy-efficient way. To do so, plants have evolved complex mechanisms to sense and transduce environmental stimuli into adaptive responses. In addition to various sensors and receptors at the plasma membrane, intracellular organelles are recently emerging as key sensors of environmental stresses such as water limitation and pathogen attack. We still lack a profound mechanistic understanding of how organelles communicate and coordinate the complex stress responses. The Inter-organelle Stress Signalling team performs studies to understand the complex organelle-organelle and organelle-to-nucleus cross-talk using high-end multi-omics and cell biology approaches.
itochondria and chloroplasts are emerging as key environmental sensors. Upon perturbation of their metabolic functions and alteration of their redox status by stress, they communicate to the nucleus to regulate stress-responsive gene expression. We previously identified one of the first mechanisms of retrograde signaling in which organellar stress signals are communicated to the nucleus via the release of ER-membrane-bound ARABIDOPSIS NAC DOMAIN CONTAINING PROTEIN 13 and 17 transcription factors. Using ANAC013 and 17 as a model system, we aim to further deepen our understanding of the still poorly understood organelle-ER and organelle-to-nucleus signalling mechanisms using high-end interactomics and chemical proteomics.
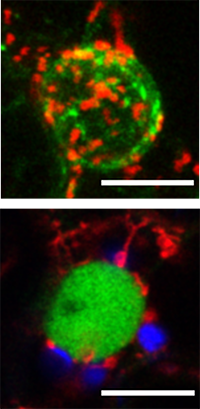
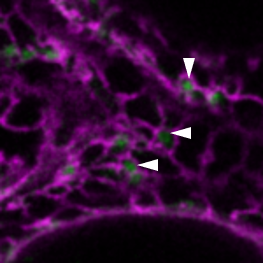
ue to their tight integration into the cell’s metabolic and signalling networks, organelles are in a prime position to communicate stress signals and coordinate optimal adaptive responses. However, we lack a profound understanding of how these organelles communicate stress signals with each other. In our lab we study a novel mechanism of communication between organelles, more specifically, by their direct physical association, through the identification of stress-induced contact sites with the use of state-of-the-art live-cell imaging and proteomics methods. Although inter-organellar contact sites have been studied in animal and yeast systems, nearly nothing is known on their existence and mode-of-action in plants. The results of the project aim a better understanding and re-evaluation of the fundamental mechanisms of plant responses and adaptation to stress.
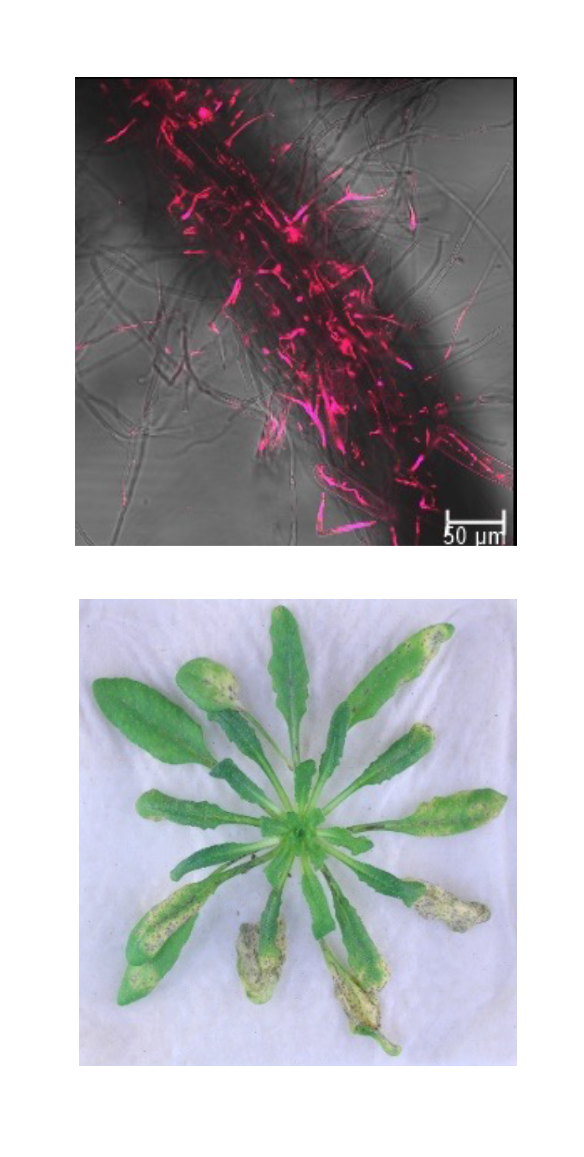
In order to obtain specific and sustainable solutions for crop protection, we need a better understanding of the molecular interactions between plants and phytopathogens. Intracellular organelles such as chloroplasts and mitochondria play a pivotal role in plant immune responses. Our team aims to identify pathogen-secreted effector proteins that target the plant host organelles. Together with the identification of the specific target proteins within the host organelles, we aim a better insight at the molecular level into plant-pathogen interactions and the involvement of organelles in plant immunity.